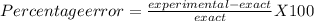Chemistry, 21.03.2020 22:38 imjustdumbsis
The exact value for the density of aluminum is 2.699 g. cm^3. Working in the science lab at school, Joseph finds the density of a piece of aluminum to be 2.75 g/cm^3. What is Joseph's percent error? (Round to the nearest hundredth.)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1


You know the right answer?
The exact value for the density of aluminum is 2.699 g. cm^3. Working in the science lab at school,...
Questions


English, 12.07.2019 10:00




Mathematics, 12.07.2019 10:00


English, 12.07.2019 10:00


English, 12.07.2019 10:00

Chemistry, 12.07.2019 10:00


Social Studies, 12.07.2019 10:00




Mathematics, 12.07.2019 10:00

Mathematics, 12.07.2019 10:00


Biology, 12.07.2019 10:00