
Chemistry, 21.03.2020 20:28 kragland4752
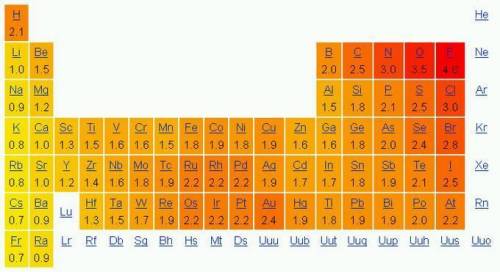
Use the electronegativities in the periodic table above to determine which bonding pair forms polar covalent bonds.
A) Lithium and Oxygen
B) Calcium and Chlorine
C) Nitrogen and Hydrogen
D) Hydrogen and Hydrogen

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Which of the following statements concerning the influence of culture on ethnic identity formation is accurate? a. one will reject ethnic identity if cultural stereotypes are encountered. b. if one’s ethnic city is different from the dominant cultural group, then one’s ethnic identity you will become weekend. c. if an the ethnic group is excepted by dominant culture, then ethnic identity formation can be a difficult process. d. similarity to the dominant culture can determine how easy it is for one to except ethnic differences.
Answers: 2

Chemistry, 22.06.2019 03:10
The covalent compound acetylene, which is the fuel of the oxyacetylene torch used by welders, has the molecular formula c2h2. the covalent compound benzene, a commercial solvent, has the molecular formula c6h6 each of these covalent compounds contains carbon and hydrogen atoms in a one-to-one ratio. would it be correct to write the chemical formulas of each as ch? explain.
Answers: 1

Chemistry, 22.06.2019 23:30
Why do oxygen have a strong attractive force for electrons
Answers: 2

Chemistry, 23.06.2019 01:30
Adirect relationship can be represented by: a curve a pie chart
Answers: 2
You know the right answer?
Use the electronegativities in the periodic table above to determine which bonding pair forms polar...
Questions


Physics, 27.01.2022 14:00


Biology, 27.01.2022 14:00

Mathematics, 27.01.2022 14:00

Chemistry, 27.01.2022 14:00

Social Studies, 27.01.2022 14:00


English, 27.01.2022 14:00


Mathematics, 27.01.2022 14:00


Mathematics, 27.01.2022 14:00

English, 27.01.2022 14:00

Mathematics, 27.01.2022 14:00

Mathematics, 27.01.2022 14:00


Biology, 27.01.2022 14:00


Mathematics, 27.01.2022 14:00



