
Chemistry, 21.03.2020 11:31 brenda2512
The following three equations represent reactions done in this lab exercise. Classify each reaction by their type. Note there may be more than one way to classify a reaction
1. Cu(s)+4HNO3(aq)⟶Cu(NO3)2(aq)+2NO2(g )+2H2O(l)
a. precipitation.
b. double displacement.
c. decomposition.
d. single displacement.
e. oxidation / reduction.
f. synthesis / combination.
2. Cu(NO3)2(aq)+2NaOH(aq)⟶Cu(OH)2(s)+2 NaNO3(aq)
a. synthesis / combination.
b. single displacement.
c. oxidation / reduction.
d. decomposition.
e. double displacement.
f. precipitation.
3. CuO(s)+2HCl(aq)⟶CuCl2(aq)+H2O(l)
a. precipitation.
b. double displacement.
c. synthesis / combination.
d. oxidation / reduction.
e. single displacement.
f. decomposition.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 07:10
Remember to use the proper number of significant figures and leading zeros in all calculations.gelatin has a density of 1.27 g/cm³. if you have a blob of gelatin dessert that fills a 2.0 liter bottle, what is its mass? 2540 g2500 g3.9 x 10-43.937x 10-4
Answers: 3

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

You know the right answer?
The following three equations represent reactions done in this lab exercise. Classify each reaction...
Questions


Mathematics, 05.07.2019 15:00


History, 05.07.2019 15:00

Mathematics, 05.07.2019 15:00



Mathematics, 05.07.2019 15:00


Mathematics, 05.07.2019 15:00





History, 05.07.2019 15:00


History, 05.07.2019 15:00



Chemistry, 05.07.2019 15:00

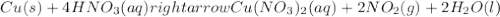 : oxidation reduction
: oxidation reduction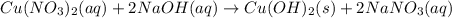 : precipitation
: precipitation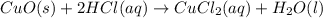 : Double displacement
: Double displacement

