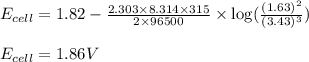A galvanic cell at a temperature of 42 degrees Celcius is powered by the following redox reaction:
3CU2+(aq) + 2Al(s) > 3Cu(s) + 2Al3+(aq)
Suppose the cell is prepared with 3.43 M Cu2+n one half-cell and 1.63 M Al3+in the other.
A) Calculate the cell voltage under these conditions. Round your answer to 3 significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 17:00
Reduction is a reaction which results in a in electrons and a in positive charge of the atom or ion 1) a- loss 1) b- gain 2) a-increase 2) b-decrease
Answers: 1

Chemistry, 22.06.2019 17:20
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2


Chemistry, 23.06.2019 03:00
You have a sample of a metal, the sample is exactly 6.02 x 1023atom, if the sample has a mass 55.85 what metal is your sample made of?
Answers: 2
You know the right answer?
A galvanic cell at a temperature of 42 degrees Celcius is powered by the following redox reaction:
Questions


English, 09.12.2020 21:40



Mathematics, 09.12.2020 21:40


Mathematics, 09.12.2020 21:40



Mathematics, 09.12.2020 21:40



Mathematics, 09.12.2020 21:40

Mathematics, 09.12.2020 21:40



English, 09.12.2020 21:40



Biology, 09.12.2020 21:40

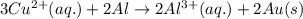
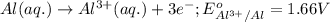 ( × 2)
( × 2)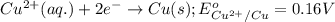 ( × 3)
( × 3)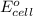 of the reaction, we use the equation:
of the reaction, we use the equation: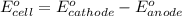
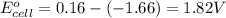
![E_{cell}=E^o_{cell}-\frac{2.303RT}{nF}\log \frac{[Al^{3+}]^2}{[Cu^{2+}]^3}](/tpl/images/0557/5138/3aff8.png)
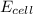 = electrode potential of the cell = ? V
= electrode potential of the cell = ? V![42^oC=[42+273]K=315K](/tpl/images/0557/5138/563a7.png)
![[Al^{3+}]=1.63M](/tpl/images/0557/5138/1e5e1.png)
![[Cu^{2+}]=3.43M](/tpl/images/0557/5138/455ea.png)