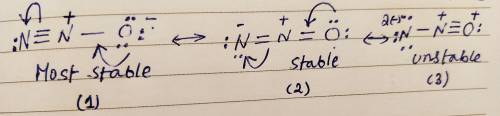Chemistry, 21.03.2020 03:16 winterblanco
Draw three resonance structures for N2O. This species has its three atoms bonded sequentially in the following fashion: N-O. Draw your resonance structures so that the atoms in them are bonded together in this order. Select the most important resonance structure for this species based on the formal charges on the atoms of the three resonance structures you have drawn. Now select the statement from the multiple choices which is true about this most important resonance structure. In the most important resonance structure of N2O :

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1


You know the right answer?
Draw three resonance structures for N2O. This species has its three atoms bonded sequentially in the...
Questions

Mathematics, 10.03.2021 07:00



Mathematics, 10.03.2021 07:00

History, 10.03.2021 07:00

History, 10.03.2021 07:00

Mathematics, 10.03.2021 07:00


Mathematics, 10.03.2021 07:00


History, 10.03.2021 07:00

Mathematics, 10.03.2021 07:00

History, 10.03.2021 07:00



Mathematics, 10.03.2021 07:00

Mathematics, 10.03.2021 07:00

Mathematics, 10.03.2021 07:00

Mathematics, 10.03.2021 07:00