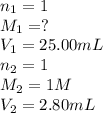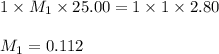Chemistry, 21.03.2020 03:13 ellisc7044
1. Determine the volume of 1M NaOH that is required to reach the equivalence point with 25.00 mL of HCl (of unknown concentration). From there, calculate the original concentration of the unknown HCl solution.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:10
3.) for each of the following compounds, draw the major organic product of reaction with hcl or naoh and circle whether the starting materials and products will be more soluble in organic solvent or water benzoic acid + hcl: benzoic acid + naoh: oh benzoic acid water/organic water organic fluorenone hс: fluorenone + naoh: fluorenone water/organic water/organic веnzocaine + hci: benzocaine + n»oh: h2n benzocaine water/organic water organic o=
Answers: 3

Chemistry, 22.06.2019 05:30
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3

Chemistry, 22.06.2019 14:00
Which of the following is true about a carbonated soft drink? . the carbon dioxide is the solvent, and water is the solute.. the water is the solution, and carbon dioxide is the solvent.. the carbon dioxide is the solution, and the water is the solvent.. the water is the solvent, and the carbon dioxide is the solute.. .
Answers: 1

Chemistry, 22.06.2019 22:30
Which is a characteristic of the electron sea model for metallic bonding? molecular orbitals overlap to produce bands. electrons flow easily between metal nuclei. electrons are in fixed positions in the orbitals. atomic nuclei are arranged in an irregular pattern.
Answers: 3
You know the right answer?
1. Determine the volume of 1M NaOH that is required to reach the equivalence point with 25.00 mL of...
Questions

Mathematics, 05.07.2021 19:10


Mathematics, 05.07.2021 19:10






Computers and Technology, 05.07.2021 19:10


Computers and Technology, 05.07.2021 19:10


World Languages, 05.07.2021 19:10

Mathematics, 05.07.2021 19:10

Mathematics, 05.07.2021 19:10



Mathematics, 05.07.2021 19:20



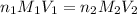
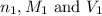 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 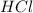
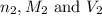 are the n-factor, molarity and volume of base which is NaOH.
are the n-factor, molarity and volume of base which is NaOH.