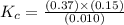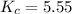Chemistry, 21.03.2020 02:58 alexisfaithsmith
At the equilibrium point in the decomposition of phosphorus pentachloride to chlorine and phosphorus trichloride, the following concentrations are obtained: 0.010 mol/L PCl5, 0.15 mol/l PCl3 and 0.37 mol/L Cl2. Determine the Keq for the reaction

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1


Chemistry, 23.06.2019 04:00
Achemical reaction is done in the setup shown , resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 2

Chemistry, 23.06.2019 04:10
An unknown substance has been shown to have weak covalent bonds. which of the following is most likely a property of this substance? a. high ph b. high conductivity c. low melting point d. low flammability
Answers: 3
You know the right answer?
At the equilibrium point in the decomposition of phosphorus pentachloride to chlorine and phosphorus...
Questions


Mathematics, 16.10.2020 03:01





Business, 16.10.2020 03:01

Mathematics, 16.10.2020 03:01






Chemistry, 16.10.2020 03:01



Mathematics, 16.10.2020 03:01

Mathematics, 16.10.2020 03:01


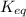 for the reaction is 5.55
for the reaction is 5.55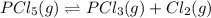
![K_c=\frac{[Cl_2]\times [PCl_3]}{[PCl_5]}](/tpl/images/0557/1142/ffe89.png)