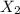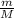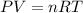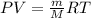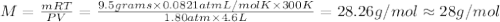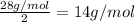To identify a diatomic gas (X2), a researcher carried out the following experiment: She weighed an empty 4.6-L bulb, then filled it with the gas at 1.80 atm and 27.0 ∘C and weighed it again. The difference in mass was 9.5 g . Identify the gas. Express your answer as a chemical formula. View Available Hint(s)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which type of bonding involves the complete transfer of a valence electron from a less electrogrative atom to a more electronegative one
Answers: 1

Chemistry, 22.06.2019 10:00
Americium-241 undergoes fission to produce three neutrons per fission event. if a neutron-absorbing material is mixed in with this sample so that the rate of neutron production drops down to 1.8 neutrons per fission event, which will be effective at achieving a critical mass? check all that apply. remove a deflective shield surrounding the sample. remove absorbent material mixed in with the sample. compress the sample of americium-241.
Answers: 1

Chemistry, 22.06.2019 14:30
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3

You know the right answer?
To identify a diatomic gas (X2), a researcher carried out the following experiment: She weighed an e...
Questions





Physics, 24.07.2019 22:00

Mathematics, 24.07.2019 22:00


Physics, 24.07.2019 22:00




Mathematics, 24.07.2019 22:00



History, 24.07.2019 22:00



Chemistry, 24.07.2019 22:00


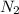 .
.