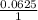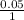Chemistry, 20.03.2020 12:03 j1theking18
When aqueous solutions of Na2SO4 and Pb(NO3)2 are mixed, PbSO4 precipitates. Calculate the mass of PbSO4 formed when 1.25 L of 0.0500 M Pb(NO3)2 and 2.00 L of 0.0250 M Na2SO4 are mixed.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:30
What is a scientific theory? a. a scientist's guess about how something works b. the results of an experiment obtained using the scientific method c. a proven fact that will never change d. an idea that is backed by data from many sources
Answers: 2


Chemistry, 22.06.2019 05:00
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3

Chemistry, 22.06.2019 10:30
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1
You know the right answer?
When aqueous solutions of Na2SO4 and Pb(NO3)2 are mixed, PbSO4 precipitates. Calculate the mass of P...
Questions



Mathematics, 05.11.2020 19:30


Mathematics, 05.11.2020 19:30

Chemistry, 05.11.2020 19:30

Advanced Placement (AP), 05.11.2020 19:30

Spanish, 05.11.2020 19:30

Chemistry, 05.11.2020 19:30

Engineering, 05.11.2020 19:30


English, 05.11.2020 19:30

Mathematics, 05.11.2020 19:30



Mathematics, 05.11.2020 19:30



Mathematics, 05.11.2020 19:30