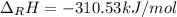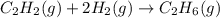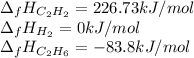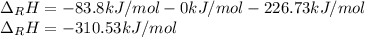Chemistry, 20.03.2020 12:02 tae8002001
Hydrogenation of double and triple bonds is an important industrial process. Calculate (in kJ/mole) the standard enthalpy change ΔH° for the hydrogenation of ethyne (acetylene) to ethane. Just enter a number (no units).

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:10
Describe the difference between a. a hypothesis and a theory and b. an observation and an experiment.
Answers: 1

Chemistry, 22.06.2019 13:30
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1

Chemistry, 22.06.2019 23:00
Movement that is like a t a type of wave that transfers energy where the particles in the medium move in a circle motion while the energy travels left or right. a type of wave that transfers energy where the particles in the medium move perpendicular to the direction in which the energy is traveling. transfers energy from one location to another a type of wave that transfers energy where the particles in the medium move parallel to the direction in which the energy is traveling. movement that is back and forth, like an equal sign = 1. wave 2. parallel movement 3. perpendicular movement 4. transverse wave 5. longitudinal wave 6. surface wave
Answers: 1
You know the right answer?
Hydrogenation of double and triple bonds is an important industrial process. Calculate (in kJ/mole)...
Questions

Social Studies, 23.10.2019 16:00

Mathematics, 23.10.2019 16:00


Mathematics, 23.10.2019 16:00



History, 23.10.2019 16:00




Mathematics, 23.10.2019 16:00

Social Studies, 23.10.2019 16:00

Chemistry, 23.10.2019 16:00

Biology, 23.10.2019 16:00

English, 23.10.2019 16:00




Mathematics, 23.10.2019 16:00

Social Studies, 23.10.2019 16:00