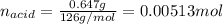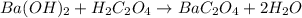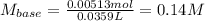Chemistry, 20.03.2020 11:54 jadenpmoore2008
Oxalic acid dihydrate is a solid, diprotic acid that can be used in the laboratory as a primary standard. Its formula is H2C2O4•2H2O. A student dissolves 0.647 grams of H2C2O4•2H2O in water and titrates the resulting solution with a solution of barium hydroxide of unknown concentration. If 35.9 mL of the barium hydroxide solution are required to neutralize the acid, what is the molarity of the barium hydroxide solution ?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 12:30
Type the correct answer in the box. spell all words correctly .what does biodiesel produce in higher amounts? biodiesel produces higher amounts
Answers: 2

Chemistry, 21.06.2019 14:10
Which of these will change if the air in aclosed bottle is heated? abcdthe mass of the airthe composition of the airthe air pressure in the bottlethe number of air molecules in the bottle
Answers: 3

Chemistry, 22.06.2019 00:30
Sarah wants to know where in her garden chamomile would grow the best. she thinks chamomile will grow best in the corner of the garden that gets the most sunlight. to test her hypothesis, she decides to plant several groups of chamomile in her garden as an experiment. which of the following variables will sarah need to measure to know which group of plants grew best? a. the location of the plants b. the type of plants c. the height of the plants d. the amount of water she gives the plants
Answers: 1

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3
You know the right answer?
Oxalic acid dihydrate is a solid, diprotic acid that can be used in the laboratory as a primary stan...
Questions



Mathematics, 18.02.2021 02:30




Biology, 18.02.2021 02:30



Mathematics, 18.02.2021 02:30

Chemistry, 18.02.2021 02:30

Mathematics, 18.02.2021 02:30

Mathematics, 18.02.2021 02:30


Mathematics, 18.02.2021 02:30



Mathematics, 18.02.2021 02:30


Mathematics, 18.02.2021 02:30