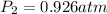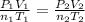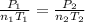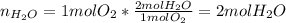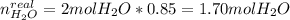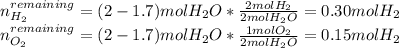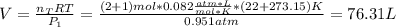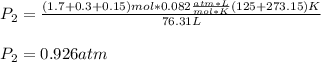A mixture in which the mole ratio of hydrogen to oxygen is (exactly) 2:1 is used to prepare water by the reaction 2 H2 (g) + O2 (g) → 2 H2O (g) The total pressure in the container is 0.951 atm at 22°C before the reaction. What is the final pressure in the container after the reaction, with a final temperature of 125°C, no volume change, and an 85.0% yield?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3


Chemistry, 22.06.2019 18:30
When a device is used in a circuit in which the voltage is 81 v the current flowing through the device is 3 a what is the resistance of the device
Answers: 2

Chemistry, 22.06.2019 20:00
State one important difference between a physical change and a chemical change?
Answers: 1
You know the right answer?
A mixture in which the mole ratio of hydrogen to oxygen is (exactly) 2:1 is used to prepare water by...
Questions

Mathematics, 18.09.2020 07:01

Physics, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

English, 18.09.2020 07:01

History, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

Mathematics, 18.09.2020 08:01

Mathematics, 18.09.2020 08:01

Mathematics, 18.09.2020 08:01

Social Studies, 18.09.2020 08:01

Geography, 18.09.2020 08:01

Mathematics, 18.09.2020 08:01

Mathematics, 18.09.2020 08:01

Arts, 18.09.2020 08:01

Mathematics, 18.09.2020 08:01

English, 18.09.2020 08:01

Geography, 18.09.2020 08:01

English, 18.09.2020 08:01

Mathematics, 18.09.2020 08:01