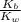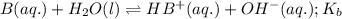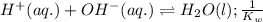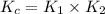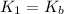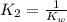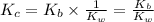Chemistry, 20.03.2020 10:23 briseno138
Given the following equilibrium constants: Kb B(aq) + H2O(l) ⇌ HB+(aq) + OH−(aq) 1/Kw H+(aq) + OH−(aq) ⇌ H2O(l) What is the equilibrium constant for the following reaction equal to? B(aq) + H+(aq) ⇌ HB+(aq)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 13:30
A48 g piece of ice at 0.0 ∘c is added to a sample of water at 7.4 ∘c. all of the ice melts and the temperature of the water decreases to 0.0 ∘c. how many grams of water were in the sample?
Answers: 1

Chemistry, 21.06.2019 22:00
Fission of uranium-235 products energy and a. isotopes of smaller elements b. isotopes of larger elements c. lighter isotopes of uranium d. heavier isotopes of uranium
Answers: 3

Chemistry, 22.06.2019 14:00
What term describes technology that operates on an atomic level
Answers: 2

Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1
You know the right answer?
Given the following equilibrium constants: Kb B(aq) + H2O(l) ⇌ HB+(aq) + OH−(aq) 1/Kw H+(aq) + OH−(a...
Questions

Arts, 01.06.2021 18:10


Mathematics, 01.06.2021 18:10


English, 01.06.2021 18:10

Biology, 01.06.2021 18:10



Mathematics, 01.06.2021 18:10

Mathematics, 01.06.2021 18:10

Social Studies, 01.06.2021 18:10

History, 01.06.2021 18:10



Mathematics, 01.06.2021 18:10


Mathematics, 01.06.2021 18:10

Biology, 01.06.2021 18:10

Mathematics, 01.06.2021 18:10

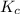 for the net reaction is
for the net reaction is