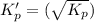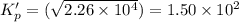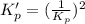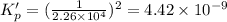Chemistry, 20.03.2020 09:47 jessicamcummins
This reaction has an equilibrium constant of Kp = 2.26 * 104 at 298 K: CO(g) + 2 H2(g) ∆ CH3OH(g) Calculate Kp for each reaction and predict whether reactants or products will be favored at equilibrium:

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:40
If the atomic mass of an atom is 34 and the atom contains 13 protons, how many neutrons does the atom contain?
Answers: 2

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2


Chemistry, 22.06.2019 23:00
Condensation happens when water vapor cools. true or false? ?
Answers: 2
You know the right answer?
This reaction has an equilibrium constant of Kp = 2.26 * 104 at 298 K: CO(g) + 2 H2(g) ∆ CH3OH(g) Ca...
Questions




Business, 18.10.2020 09:01


Mathematics, 18.10.2020 09:01

Mathematics, 18.10.2020 09:01



Mathematics, 18.10.2020 09:01

History, 18.10.2020 09:01

Mathematics, 18.10.2020 09:01

Mathematics, 18.10.2020 09:01





Social Studies, 18.10.2020 09:01

Social Studies, 18.10.2020 09:01

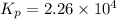 at 298K.
at 298K.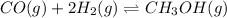
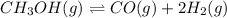
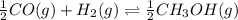
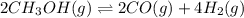
 : reactants are favoured
: reactants are favoured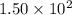 : products are favoured
: products are favoured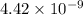 : reactants are favoured
: reactants are favoured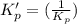
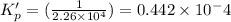
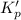 is less than 1, reactants will be favored at equilibrium.
is less than 1, reactants will be favored at equilibrium.