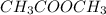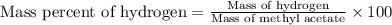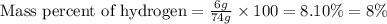Chemistry, 20.03.2020 09:48 KennyMckormic
This is the chemical formula for methyl acetate: CH32CO2. Calculate the mass percent of hydrogen in methyl acetate. Round your answer to the nearest percentage. %

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 07:30
Plz mark brainliest 30 points 1) find the momentum of a 12 kg snowball that is rolling with a velocity of 9 m/s. 2) an 8 ball with a mass of .5 kg is sitting at rest. it is hit by the cue ball (1 kg) traveling at 2.5 m/s. if the cue ball is at rest after the collision, how fast is the 8 ball traveling after the collision? 3) two football players are running toward each other. if the offensive player is 75 kg and is running 8 m/s, how fast must the 60 kg defensive player run in order for the two players to hit and stop?
Answers: 1

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 09:00
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1
You know the right answer?
This is the chemical formula for methyl acetate: CH32CO2. Calculate the mass percent of hydrogen in...
Questions

Physics, 15.10.2020 07:01

Physics, 15.10.2020 07:01


Mathematics, 15.10.2020 07:01

History, 15.10.2020 07:01

Health, 15.10.2020 07:01


Computers and Technology, 15.10.2020 07:01

Mathematics, 15.10.2020 07:01

Mathematics, 15.10.2020 07:01


Mathematics, 15.10.2020 07:01

Mathematics, 15.10.2020 07:01


Computers and Technology, 15.10.2020 07:01


Mathematics, 15.10.2020 07:01

Geography, 15.10.2020 07:01

Physics, 15.10.2020 07:01

Mathematics, 15.10.2020 07:01