
Chemistry, 20.03.2020 08:30 darwin59651
Calculate the pH of a 0.437 M aqueous solution of hydrocyanic acid (HCN, Ka = 4.0×10-10) and the equilibrium concentrations of the weak acid and its conjugate base.

Answers: 2
Another question on Chemistry



Chemistry, 22.06.2019 23:30
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1

Chemistry, 23.06.2019 00:00
What does an electron configuration for an atom relate to the atoms placement on the periodic table
Answers: 2
You know the right answer?
Calculate the pH of a 0.437 M aqueous solution of hydrocyanic acid (HCN, Ka = 4.0×10-10) and the equ...
Questions

Social Studies, 12.07.2019 12:50


Chemistry, 12.07.2019 12:50


Mathematics, 12.07.2019 13:00


Mathematics, 12.07.2019 13:00



Mathematics, 12.07.2019 13:00


History, 12.07.2019 13:00





Business, 12.07.2019 13:00

Biology, 12.07.2019 13:00


![[HCN]_{eq}=0.43699M](/tpl/images/0555/6850/8a21f.png)
![[CN^-]_{eq}=1.322x10^{-5}M](/tpl/images/0555/6850/83c77.png)
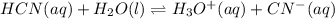
![Ka=\frac{[H^+]_{eq}[CN^-]_{eq}}{[HCN]_{eq}}](/tpl/images/0555/6850/4425b.png)
 due to the reaction extent, goes:
due to the reaction extent, goes: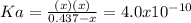
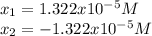
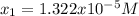
![pH=-log([H^+])=-log(1.322x10^{-5})=4.88](/tpl/images/0555/6850/f810b.png)
![[HCN]_{eq}=0.437M-1.322x10^{-5}M=0.43699M](/tpl/images/0555/6850/d4f3f.png)


