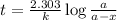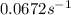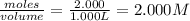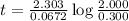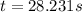At a certain fixed temperature, the reaction A(g) + 2 B(g) → AB2(g) is found to be first order in the concentration of A and zero order in the concentration of B. The reaction rate constant is 0.0672 s−1 . If 2.000 moles of A and 4.000 moles of B are placed in a 1.000 liter container, how many seconds will elapse before the concentration of A has fallen to 0.300 mol/liter? 1. 36.6948 2. 60.226 3. 63.2373 4. 34.244 5. 49.9242 6. 23.8631 7. 51.2735 8. 29.6425 9. 36.1356 10. 28.231 Answer in units of s.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 22.06.2019 22:30
Which of the following is true about the speed of light? it depends on the wavelength.
Answers: 3
You know the right answer?
At a certain fixed temperature, the reaction A(g) + 2 B(g) → AB2(g) is found to be first order in th...
Questions

Mathematics, 23.05.2021 22:10

History, 23.05.2021 22:10

Chemistry, 23.05.2021 22:10

Mathematics, 23.05.2021 22:10

Mathematics, 23.05.2021 22:10








Advanced Placement (AP), 23.05.2021 22:10

Mathematics, 23.05.2021 22:10


Mathematics, 23.05.2021 22:10


History, 23.05.2021 22:10


History, 23.05.2021 22:10

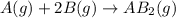
![Rate=k[A]^1[B]^0](/tpl/images/0555/2030/f8c47.png)