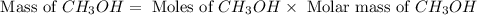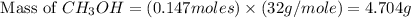Based on the ideal gas law, there is a simple equivalency that exists between the amount of gas and the volume it occupies. At standard temperature and pressure (STP; 273.15 K and 1 atm, respectively), one mole of gas occupies 22.4 L of volume. What mass of methanol (CH3OH) could you form if you reacted 6.59 L of a gas mixture (at STP) that contains an equal number of carbon monoxide (CO) and hydrogen gas (H2) molecules?

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2

Chemistry, 22.06.2019 16:00
Answer asap : ( a. how does mucus prevent the entry of pathogens? b. describe two ways white blood cells protect us from pathogens.
Answers: 1

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1
You know the right answer?
Based on the ideal gas law, there is a simple equivalency that exists between the amount of gas and...
Questions


History, 18.12.2020 19:50


Mathematics, 18.12.2020 19:50











Mathematics, 18.12.2020 19:50

Chemistry, 18.12.2020 19:50


Mathematics, 18.12.2020 19:50

Mathematics, 18.12.2020 19:50

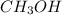 produced is, 4.704 grams.
produced is, 4.704 grams.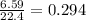 mole of gas mixture
mole of gas mixture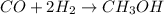
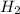 react with 1 mole of
react with 1 mole of 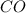
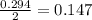 moles of
moles of