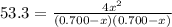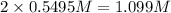Chemistry, 19.03.2020 23:27 shermoisllo3
At a certain temperature, the equilibrium constant, K c , Kc, for this reaction is 53.3. H 2 ( g ) + I 2 ( g ) − ⇀ ↽ − 2 HI ( g ) K c = 53.3 H2(g)+I2(g)↽−−⇀2HI(g)Kc=53.3 At this temperature, 0.700 mol H 2 0.700 mol H2 and 0.700 mol I 2 0.700 mol I2 were placed in a 1.00 L container to react. What concentration of HI HI is present at equilibrium?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Plz me get these answer dubble cheak ur answer plz ppl i need it right
Answers: 2

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 13:00
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2
You know the right answer?
At a certain temperature, the equilibrium constant, K c , Kc, for this reaction is 53.3. H 2 ( g ) +...
Questions


History, 02.09.2019 00:30

History, 02.09.2019 00:30


Mathematics, 02.09.2019 00:30

History, 02.09.2019 00:30


Social Studies, 02.09.2019 00:30




Mathematics, 02.09.2019 00:30



Mathematics, 02.09.2019 00:30




Mathematics, 02.09.2019 00:30

Mathematics, 02.09.2019 00:30

![[H_2]=\frac{0.700 mol}{1.00 L}=0.700 M](/tpl/images/0554/8381/9fcad.png)
![[I_2]=\frac{0.700 mol}{1.00 L}=0.700 M](/tpl/images/0554/8381/1f00e.png)
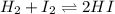
![K_c=\frac{[HI]^2}{[H_2][I_2]}](/tpl/images/0554/8381/62646.png)