Chemistry, 19.03.2020 21:25 discolldiane7351
A sample of hexane (C6H14) has a mass of 0.580 g. The sample is burned in a bomb calorimeter that has a mass of 1.900 kg and a specific heat of 3.21 J/giK. What amount of heat is produced during the combustion of hexane if the temperature of the calorimeter increases by 4.542 K? Use .

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:20
If i can still dissolve more sugar into the solution at a certain temperature what would i call that solution
Answers: 3

Chemistry, 22.06.2019 06:30
Design techniques and materials that reduce the negative environmental impact of a structure are referred to as
Answers: 2

Chemistry, 22.06.2019 09:00
Ineed to find the answer of this question because i dont understand it
Answers: 1

Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2
You know the right answer?
A sample of hexane (C6H14) has a mass of 0.580 g. The sample is burned in a bomb calorimeter that ha...
Questions

Mathematics, 26.02.2021 04:00




Mathematics, 26.02.2021 04:00


Mathematics, 26.02.2021 04:00




Mathematics, 26.02.2021 04:00

History, 26.02.2021 04:00

Mathematics, 26.02.2021 04:00



History, 26.02.2021 04:00

Mathematics, 26.02.2021 04:00



Physics, 26.02.2021 04:00

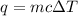
 = change in temperature = 4.542 K
= change in temperature = 4.542 K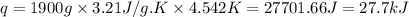
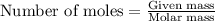
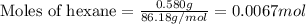
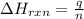
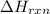 = enthalpy change of the reaction
= enthalpy change of the reaction