According to the Bronsted-Lowry definition,
an acid is a proton acceptor
a b...

Chemistry, 19.03.2020 21:01 gokusupersaiyan12345
According to the Bronsted-Lowry definition,
an acid is a proton acceptor
a base is a proton donor
a base produces H+ ions in aqueous solutions
a base is a proton acceptor

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2

Chemistry, 22.06.2019 07:20
Which of these conditions most likely produces an unstable isotope?
Answers: 1


You know the right answer?
Questions

English, 05.09.2020 03:01

Mathematics, 05.09.2020 03:01





English, 05.09.2020 03:01



Mathematics, 05.09.2020 03:01






English, 05.09.2020 03:01


English, 05.09.2020 03:01

Advanced Placement (AP), 05.09.2020 03:01

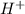 + Conjugate base
+ Conjugate base

