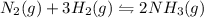Chemistry, 19.03.2020 20:43 lovelylife7553
The reaction for the synthesis of ammonia N2(g) + 3 H2(g) → 2 NH3(g) is exothermic. Increasing the temperature applied to the system I) increases the amount of NH3. II) decreases the amount of NH3. III) changes the value of Keq. IV) does not change the value of Keq.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Oxidation-reduction reactions (often called "redox" for short) are reactions that involve the transfer of electrons from one species to another. oxidation states, or oxidation numbers, allow chemists to keep track of these electron transfers. in general, one element will lose electrons (oxidation), with the result that it will increase in oxidation number, and another element will gain electrons (reduction), thereby decreasing in oxidation number. the species that is oxidized is called the reducing agent or reductant. the species that is reduced is called the oxidizing agent or oxidant. to sum up: oxidation = increase in oxidation state = loss of electrons = reducing agent reduction = decrease in oxidation state = gain of electrons = oxidizing agent part a which element is oxidized in this reaction? fe2o3+3co→2fe+3co2 enter the elemental symbol. view available hint(s) is oxidized part b which element is reduced in this reaction? 2hcl+2kmno4+3h2c2o4→6co2+2mno2+2kcl+4h2o enter the elemental symbol. view available hint(s) is reduced
Answers: 1

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 22.06.2019 13:50
Amap that uses a range of colors and shading to represent the elevation, depth, or landscape of specific features on earth is a/an map.
Answers: 3

Chemistry, 22.06.2019 16:30
Explain in detail of the four major scientific developments that spurred the formulation of the plate tectonics theory
Answers: 2
You know the right answer?
The reaction for the synthesis of ammonia N2(g) + 3 H2(g) → 2 NH3(g) is exothermic. Increasing the t...
Questions



Health, 24.10.2019 10:43

Mathematics, 24.10.2019 10:43




History, 24.10.2019 10:43

Health, 24.10.2019 10:43

Advanced Placement (AP), 24.10.2019 10:43

Mathematics, 24.10.2019 10:43


English, 24.10.2019 10:43

Social Studies, 24.10.2019 10:43


Mathematics, 24.10.2019 10:43

Mathematics, 24.10.2019 10:43

Spanish, 24.10.2019 10:43