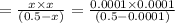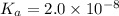Chemistry, 19.03.2020 20:40 afosburgh20
50 POINTS PLEASE HELP ASAP! I WILL MARK BRAINLIEST ONLY IF CORRECT AND CLEARLY STATED
19. Calculate the hydrogen-ion concentration [H^+] for the aqueous solution in which [OH^-] is 1 x 10^-11 mol/L. Is this solution acidic, basic, or neutral? Show your work. (3 points)
20. Calculate the acid dissociation constant of a weak monoprotic acid if a 0.5 M solution of this acid gives a hydrogen-ion concentration of 0.000 1.M? Show your work. (3 points)
DO NOT Copy and past the work from another answer UNLESS you can clearly state all the steps. I've seen them all so I know If you copy.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:30
List four observations that indicate that a chemical reaction may be taking place
Answers: 1

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 13:30
Apush or pull that moves or changes and object when to objects touch
Answers: 2
You know the right answer?
50 POINTS PLEASE HELP ASAP! I WILL MARK BRAINLIEST ONLY IF CORRECT AND CLEARLY STATED
1...
1...
Questions






Advanced Placement (AP), 13.11.2019 22:31


Computers and Technology, 13.11.2019 22:31


Social Studies, 13.11.2019 22:31

Mathematics, 13.11.2019 22:31







Social Studies, 13.11.2019 22:31


Mathematics, 13.11.2019 22:31

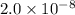 .
.![[OH^-]=1\times 10^{-11}](/tpl/images/0554/4732/291a3.png)
![pOH=-\log[OH^-]](/tpl/images/0554/4732/fe336.png)
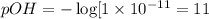
![pH=-\log[H^+]](/tpl/images/0554/4732/cf945.png)
![3=-\log[H^+]](/tpl/images/0554/4732/66d3f.png)
![[H^+]=10^{-3} M=0.001 M](/tpl/images/0554/4732/fcc5c.png)
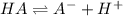
![[H^+]=x=0.0001 M](/tpl/images/0554/4732/b3468.png)
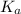 is given as:
is given as:![K_a=\frac{[A^-][H^+]}{[HA]}](/tpl/images/0554/4732/a5cb9.png)