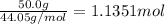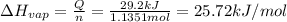Chemistry, 19.03.2020 20:37 rexerlkman4145
29.2 kJ of heat is required to convert an 50.0 g sample of
acetaldehyde from the liquid to gas phase. The molecular
weight of acetaldehyde is 44.05 g/mol. What is the heat of
vaporization of acetaldehyde in kJ/mol?
kJ/mol

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

Chemistry, 23.06.2019 03:00
In november 1987, a massive iceberg broke loose from the antartic ice mass and floated free in the ocean. the chunk of ice was estimated to be 98 mi long, 25 mi wide, and 750 ft thick. a typical backyard swimming pool contains about 24,000 gallons of water. how many of these pools could you fill from the water in this iceberg? (assume the iceberg is a rectangular solid of the above dimensions and consists of water only). express answer in scientific notation.
Answers: 1

Chemistry, 23.06.2019 04:31
Pls i will do pls imma diewhat forms white light? (4 points)a. combination of all wavelengths of ultraviolet light b. combination of all wavelengths of visible lightc. absorption of electromagnetic waves d. absorption of infrared rays
Answers: 2
You know the right answer?
29.2 kJ of heat is required to convert an 50.0 g sample of
acetaldehyde from the liquid to gas...
acetaldehyde from the liquid to gas...
Questions

English, 15.07.2019 16:30






History, 15.07.2019 16:30

Mathematics, 15.07.2019 16:30





History, 15.07.2019 16:30