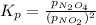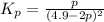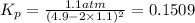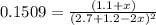Chemistry, 19.03.2020 09:36 TanishaSchollaert1
Nitrogen dioxide is one of the many oxides of nitrogen (often form another form of NOx, dinitrogen tetroxide A chemical engineer studying this reaction fils a 500. mL flask at 7.9 °C with 4.9 atm of nitrogen dioxide gas. He thèn raises the temperature considerably and when the mixture has come to equilibrium determines that it contains 2.7 atm of nitrogen dioxide gas The engineer then adds another 1.2 atm of nitrogen dioxide, and allows the mixture to come to equilibrium again. Calculate the pressure of dinitrogen tetroxide after equilibrium is reached the second time. Round your answer to 2 significant digits collectively called·N ' that are of interest to atmospheric chemistry. It can eact with ter to 囲 atm □-10 I Don't Know Submit

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 23.06.2019 01:00
What is the chemical name of the compound ti2o3? use the list of polyatomic ions and the periodic table to you answer.
Answers: 1

Chemistry, 23.06.2019 02:00
What can be done to make a solid solute dissolve faster in a liquid solvent?
Answers: 1

You know the right answer?
Nitrogen dioxide is one of the many oxides of nitrogen (often form another form of NOx, dinitrogen t...
Questions


Mathematics, 06.10.2019 12:00


Mathematics, 06.10.2019 12:00




Mathematics, 06.10.2019 12:00




Mathematics, 06.10.2019 12:00

Mathematics, 06.10.2019 12:00

Mathematics, 06.10.2019 12:00

Biology, 06.10.2019 12:00

Biology, 06.10.2019 12:00

Mathematics, 06.10.2019 12:00



Mathematics, 06.10.2019 12:00

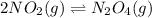
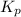 :
: