
Chemistry, 19.03.2020 08:29 RealSavage4Life
P is the pressure in atmospheres (atm), V is the volume in liters (L), n is the number of moles, R is the gas constant (0.0821 L∙atm/(mol∙K)), and T is the temperature in Kelvins (K). Consider the following conditions: a sample of neon gas was under 3.0 atm of pressure, a volume of 570 mL with a temperature of 75 °C. Assume you are going to use the ideal gas law to solve for the unknown variable. What variable are you solving for? Are all of variables in the correct units? If not, which variable needs to be converted to the correct units?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1


Chemistry, 23.06.2019 01:30
Which is an example of a highly unstable isotope that is often used in fission reactions?
Answers: 1

Chemistry, 23.06.2019 10:30
Can anyone explain 1. review your spectrometry data and use the known elements to identify the star's composition. which unknown elements make up this star? justify your element selections. 2. in parts i and ii of the lab, what happened to the electrons of each element to produce the different colors of light? explain your answers using important terms from the lesson and information provided in the laboratory. 3. stars composed of heavier (more massive) elements are often slightly older than stars made predominantly from hydrogen and helium. based on your data, is the newly discovered star a younger star? explain your answer.
Answers: 2
You know the right answer?
P is the pressure in atmospheres (atm), V is the volume in liters (L), n is the number of moles, R i...
Questions



Social Studies, 12.10.2020 02:01

Mathematics, 12.10.2020 02:01

Advanced Placement (AP), 12.10.2020 02:01

Mathematics, 12.10.2020 02:01


History, 12.10.2020 02:01

English, 12.10.2020 02:01



Mathematics, 12.10.2020 02:01


English, 12.10.2020 02:01

Chemistry, 12.10.2020 02:01

Mathematics, 12.10.2020 02:01


Computers and Technology, 12.10.2020 02:01

Mathematics, 12.10.2020 02:01

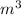
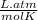
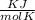
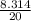 = 0.41
= 0.41 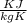
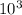 × 0.00057 = m × 0.41 × 348
× 0.00057 = m × 0.41 × 348 

