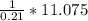A gas contains 75.0 wt% methane, 10.0% ethane, 5.0% ethylene, and the balance water. (a) Calculate the molar composition of this gas on both awet and a dry basis and the ratio (mol H2O/ mol dry gas). (b) If100kg/30%excessair,(kmol/ h)? How would the answer change if the combustion were only 75% complete?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:00
If 1.63 times 10 negative 4 of helium dissolves in 100.0g of water, what is the concentration in parts per million
Answers: 3


Chemistry, 22.06.2019 12:30
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1
You know the right answer?
A gas contains 75.0 wt% methane, 10.0% ethane, 5.0% ethylene, and the balance water. (a) Calculate t...
Questions

Mathematics, 05.03.2021 07:30


Mathematics, 05.03.2021 07:30

Mathematics, 05.03.2021 07:30


History, 05.03.2021 07:30


Mathematics, 05.03.2021 07:30

History, 05.03.2021 07:30


Mathematics, 05.03.2021 07:30



Biology, 05.03.2021 07:30


Mathematics, 05.03.2021 07:30


Mathematics, 05.03.2021 07:30

Chemistry, 05.03.2021 07:30

Mathematics, 05.03.2021 07:30

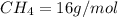
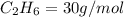
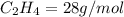

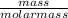
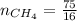
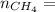 4.69 moles
4.69 moles 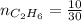
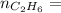 0.33 moles
0.33 moles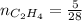
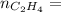 0.18 moles
0.18 moles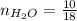
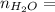 0.56 moles
0.56 moles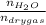
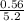




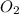 required = (2 × 4.69) + (3.5 × 0.33) + (3 × 0.18) k moles
required = (2 × 4.69) + (3.5 × 0.33) + (3 × 0.18) k moles