
Chemistry, 19.03.2020 04:08 maskythegamer
Although both N2 and 02 are naturally present in the air we breathe, high levels of NO and NO2 in the atmosphere occur mainly in regions with large automobile or power plant emissions. The equilibrium constant for the reaction of N2 and 02 to give NO is very small. The reaction is, however, highly endothermic, with a heat of reaction equal to +180 kJ (Equation 7). N2(g) + O2(g) 180 kJ 2NO(g) Equation 7 +
(a) Use LeChâtelier's Principle to explain why the concentration of NO at equilibrium increases when the reaction takes place at high temperatures.
(b) Use LeChâtelier's Principle to predict whether the concentration of NO at equilibrium should increase when the reaction takes place at high pressures.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Which compounds have the empirical formula ch2o? a.c2h4o2 b.c3h6o3 c.ch2o2 d.c5h10o5 e.c6h12o6
Answers: 3

Chemistry, 22.06.2019 05:40
Why did southern business leaders want to increase the number of slaves
Answers: 1


Chemistry, 22.06.2019 12:00
What term is applied to a scientist who studies ancient life, including animal and plant fossils a. anthropologist b. dendroclimatologist c. geophysicist d. paleontologist
Answers: 2
You know the right answer?
Although both N2 and 02 are naturally present in the air we breathe, high levels of NO and NO2 in th...
Questions

Mathematics, 06.10.2020 14:01


Mathematics, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01


Health, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01


Arts, 06.10.2020 14:01

Biology, 06.10.2020 14:01



Physics, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01

English, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01

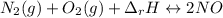
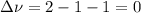
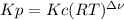
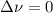 ,
, 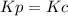 , so no effect in concentration is due to the pressure.
, so no effect in concentration is due to the pressure.

