
Consider a saturated aqueous solution of Ag2S that contains some excess solid Ag2S at the bottom. Which of the following statements is/are true? Adding AgNO3 to the solution will cause more Ag2S to dissolve. Adding some more Ag2S to the solution will cause more Ag2S to dissolve. Adding some water to the solution will cause more Ag2S to dissolve.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:50
The chemical bond connecting one nucleotide with the next one along the nucleic acid chain is called a
Answers: 3

Chemistry, 22.06.2019 14:30
Ahypothesis must be testable and falsifiable to be considered scientific a. trueb. false
Answers: 1

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 23.06.2019 02:00
Which of these is a density dependent factor? a. epidemic b. earthquake c. drought d. hurricane
Answers: 2
You know the right answer?
Consider a saturated aqueous solution of Ag2S that contains some excess solid Ag2S at the bottom. Wh...
Questions

Mathematics, 31.05.2021 04:40

Mathematics, 31.05.2021 04:40

History, 31.05.2021 04:40

Mathematics, 31.05.2021 04:40


Social Studies, 31.05.2021 04:40



Mathematics, 31.05.2021 04:40



Computers and Technology, 31.05.2021 04:40





English, 31.05.2021 04:40

World Languages, 31.05.2021 04:40

Mathematics, 31.05.2021 04:40

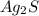 to dissolve is a true statement.
to dissolve is a true statement.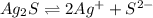
![K_{sp}=[Ag^{+}]^{2}[S^{2-}]](/tpl/images/0553/3669/0192a.png)
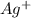 increases when
increases when 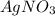 is added into solution. But value of
is added into solution. But value of 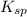 is constant at a certain temperature. Hence to keep
is constant at a certain temperature. Hence to keep 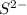 to produce more
to produce more 

