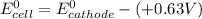A certain half-reaction has a standard reduction potential E⁰red = +0.63V. An engineer proposes using this half-reaction at the cathode of a galvanic cell that must provide at least 1.30V of electrical power. The cell will operate under standard conditions.
(a) Is there a minimum standard reduction potential that the half-reaction used at the cathode of this cell can have? If so, write "yes" and calculate the minimum. Round your answer to 2 decimal places. If there is no lower limit, write "no".
(b) Is there a maximum standard reduction potential that the half-reaction used at the cathode of this cell can have? If so, write "yes" and calculate the minimum. Round your answer to 2 decimal places. If there is no upper limit, write "no".

Answers: 3
Another question on Chemistry


Chemistry, 21.06.2019 17:30
You are performing an experiment in a lab to attempt a new method of producing pure elements from compounds. the only problem is that you do not know what element will form. by your previous calculations you know that you will have 6.3 moles of product. when it is complete, you weigh it and determine you have 604.4 grams. what element have you produced?
Answers: 1

Chemistry, 22.06.2019 05:30
Describe the interaction that occurs between two objects with the same electrical charge.
Answers: 1

You know the right answer?
A certain half-reaction has a standard reduction potential E⁰red = +0.63V. An engineer proposes usin...
Questions

Physics, 07.10.2019 20:40

Physics, 07.10.2019 20:40

Social Studies, 07.10.2019 20:40



History, 07.10.2019 20:40



Mathematics, 07.10.2019 20:40

Geography, 07.10.2019 20:40


History, 07.10.2019 20:40


English, 07.10.2019 20:40


Mathematics, 07.10.2019 20:40

Mathematics, 07.10.2019 20:40



Mathematics, 07.10.2019 20:40


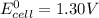
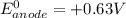
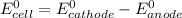
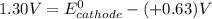
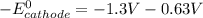
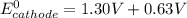
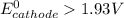 ; then
; then 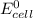 > 1.30 V
> 1.30 V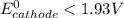 ; then
; then 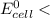 1.30 V
1.30 V