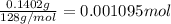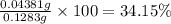Chemistry, 18.03.2020 21:26 Carlosanddana123
You are given a sample of limestone, which is mostly CaCO3, to determine the mass percentage of Ca in the rock. You dissolve the limestone in hydrochloric acid, which gives a solution of calcium chloride. Then you precipitate the calcium ion in solution by adding sodium oxalate, Na2C2O4. The precipitate is calcium oxalate, CaC2O4. You find that a sample of limestone weighing 128.3 mg gives 140.2 mg of CaC2O4. What is the mass percentage of calcium in the limestone

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

Chemistry, 22.06.2019 10:00
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1

Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2

You know the right answer?
You are given a sample of limestone, which is mostly CaCO3, to determine the mass percentage of Ca i...
Questions










English, 08.03.2021 18:30


History, 08.03.2021 18:30


Mathematics, 08.03.2021 18:30




English, 08.03.2021 18:30