
Chemistry, 18.03.2020 18:32 laceysmith2i023
Al(OH)3 + OH- → Al(OH)4- Identify whether this reaction can be classified as a Bronsted-Lowry and/or Lewis acid/base reaction. Group of answer choices Only Bronsted-Lowry Only Lewis Both Bronsted-Lowry and Lewis Neither

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 12:40
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1

Chemistry, 22.06.2019 14:30
According to le chatelier’s principle, a system in chemical equilibrium responds to stress by shifting the equilibrium in a direction that reduces the stress. normalizes the stress. increases the stress. changes the stress.
Answers: 1

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1
You know the right answer?
Al(OH)3 + OH- → Al(OH)4- Identify whether this reaction can be classified as a Bronsted-Lowry and/or...
Questions


Mathematics, 25.03.2020 18:27

Mathematics, 25.03.2020 18:28





Mathematics, 25.03.2020 18:28


Mathematics, 25.03.2020 18:28





World Languages, 25.03.2020 18:28



Mathematics, 25.03.2020 18:28

History, 25.03.2020 18:28

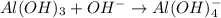
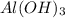 is a Lewis acid because it is an electron deficient species that means, it accepts electron pairs.
is a Lewis acid because it is an electron deficient species that means, it accepts electron pairs.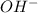 is a Lewis base because it is an electron rich species that means, it donates electron pairs.
is a Lewis base because it is an electron rich species that means, it donates electron pairs.

