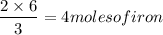Using the following equation:
Fe2O3(s) + 3 H2(g) → 2 Fe(s) + 3 H2000)
a) How many grams of...

Chemistry, 17.03.2020 22:00 holaadios222lol
Using the following equation:
Fe2O3(s) + 3 H2(g) → 2 Fe(s) + 3 H2000)
a) How many grams of iron can be made from 6 moles H2

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3


Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2

You know the right answer?
Questions


Mathematics, 30.10.2019 00:31


Mathematics, 30.10.2019 00:31

History, 30.10.2019 00:31


Advanced Placement (AP), 30.10.2019 00:31

Chemistry, 30.10.2019 00:31

Mathematics, 30.10.2019 00:31


Mathematics, 30.10.2019 00:31

Biology, 30.10.2019 00:31


Mathematics, 30.10.2019 00:31

Biology, 30.10.2019 00:31



Social Studies, 30.10.2019 00:31


Biology, 30.10.2019 00:31