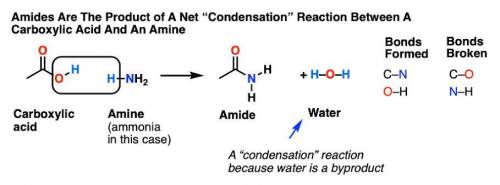
Chemistry, 17.03.2020 21:01 joycetleiji1
Draw a mechanism for the reaction of ammonia with cyclohexane carboxylic acid. In the box to the left, draw any necessary curved arrows. Show the products of the reaction in the box to the right. Include any nonzero formal charges and all lone pairs of electrons. Finally, check the box to indicate which side of the reaction is favored at equilibrium.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:30
The ph of carrots are 5.0 how it is classified a.acidic b.basic c.indicator d.neutral
Answers: 2

Chemistry, 22.06.2019 09:30
1. explain hydrogen peroxide, h 2 o 2 properties and decomposition reaction. 2. describe how each of the following natural cycles plays a part in earth’s climate system. (a) the water cycle (b) the carbon cycle
Answers: 1

Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3
You know the right answer?
Draw a mechanism for the reaction of ammonia with cyclohexane carboxylic acid. In the box to the lef...
Questions

Spanish, 18.03.2021 02:10

Mathematics, 18.03.2021 02:10



Biology, 18.03.2021 02:10



Mathematics, 18.03.2021 02:10


Mathematics, 18.03.2021 02:10


Mathematics, 18.03.2021 02:10

Social Studies, 18.03.2021 02:10


Mathematics, 18.03.2021 02:10

English, 18.03.2021 02:10

Mathematics, 18.03.2021 02:10


Social Studies, 18.03.2021 02:10

Geography, 18.03.2021 02:10