
Chemistry, 17.03.2020 20:48 sgslayerkingminecraf
A mixture of CO2 and Kr weighs 31.7 g and exerts a pressure of 0.665 atm in its container. Since Kr is expensive, you wish to recover it from the mixture. After the CO2 is completely removed by absorption with NaOH(s), the pressure in the container is 0.309 atm.
(a) How many grams of CO2 were originally present?
(b) How many grams of Kr can you recover?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1



Chemistry, 23.06.2019 01:30
Ascientist conducted an experiment and discovered that certain plants grow faster when given a particular amount of fertilizer. anouther scientist conducted the same experiment and got similar results. which concept does this best illustrate? a) repetition b) replication c) precision d) validity
Answers: 2
You know the right answer?
A mixture of CO2 and Kr weighs 31.7 g and exerts a pressure of 0.665 atm in its container. Since Kr...
Questions


Computers and Technology, 06.08.2021 17:00





Mathematics, 06.08.2021 17:00

Physics, 06.08.2021 17:00









English, 06.08.2021 17:00

Social Studies, 06.08.2021 17:00


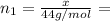
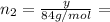
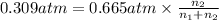
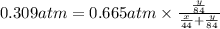
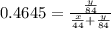 ..[2]
..[2]

