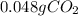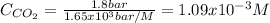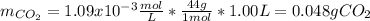Chemistry, 17.03.2020 05:24 adrian128383
How many grams of CO2 are dissolved in a 1.00 L bottle of carbonated water at 298 K if the pressure used in the carbonation process was 1.8 bar? The density of water at this temperature is 998 kg⋅m−3. The Henry's law constant for aqueous solution of CO2 at this temperature is 1.65×103bar.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 15:50
Elements in group 2 are all called alkaline earth metals. what is most similar about the alkaline earth metals?
Answers: 1

Chemistry, 23.06.2019 00:30
What is calcium oxide+diphosphorus pentoxide--> calcium phosphate balanced
Answers: 1

Chemistry, 23.06.2019 03:30
Select the correct lewis structure for fluorine which is group 7a element?
Answers: 1

Chemistry, 23.06.2019 05:00
He nucleus contains the cells genetic material in the form of dna. dna is organized into our chromosomes, which are made up of thousands of that determine our traits.
Answers: 1
You know the right answer?
How many grams of CO2 are dissolved in a 1.00 L bottle of carbonated water at 298 K if the pressure...
Questions

Mathematics, 16.12.2019 04:31

English, 16.12.2019 04:31




Spanish, 16.12.2019 04:31


Mathematics, 16.12.2019 04:31

Health, 16.12.2019 04:31

Spanish, 16.12.2019 04:31

Mathematics, 16.12.2019 04:31

Biology, 16.12.2019 04:31

Mathematics, 16.12.2019 04:31

Mathematics, 16.12.2019 04:31

Mathematics, 16.12.2019 04:31



Chemistry, 16.12.2019 04:31

French, 16.12.2019 04:31

Mathematics, 16.12.2019 04:31