

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:10
Which form of relativism states that people rely on their own standards of right and wrong when making a decision?
Answers: 1

Chemistry, 21.06.2019 23:00
Select the correct answer. given: 2libr + ba → babr2 + 2li in this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced? a. 1.18 mol b. 2.37 mol c. 4.73 mol d. 16.4 mol e. 32.9 mol
Answers: 2

Chemistry, 22.06.2019 14:40
Pastoral farming is best described as a. a method of raising livestock and moving herds b. an african method of agriculture c. a method of cultivating crops on poor soils d. a common method of desert farming select the best answer from the choices provided a b c d
Answers: 2

Chemistry, 23.06.2019 00:30
How many moles of co2 are produced during the complete combustion of 3.6 moles of c2h6
Answers: 1
You know the right answer?
A solution contains 2.0 ⨯ 10−4 M Ag+(aq) and 1.5 ⨯ 10−3 M Pb2+(aq). If NaI is added, will AgI (Ksp =...
Questions


Biology, 05.05.2020 17:31

Mathematics, 05.05.2020 17:31

Chemistry, 05.05.2020 17:31

History, 05.05.2020 17:31





Mathematics, 05.05.2020 17:31




Mathematics, 05.05.2020 17:31

English, 05.05.2020 17:31


Mathematics, 05.05.2020 17:31


History, 05.05.2020 17:31

English, 05.05.2020 17:31

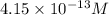 of iodide ion will result in precipitation of silver iodide.
of iodide ion will result in precipitation of silver iodide.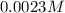 of iodide ion will result in precipitation of lead iodide.
of iodide ion will result in precipitation of lead iodide.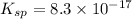
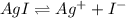
![[Ag^+]=2.0\times 10^{-4} M](/tpl/images/0549/9170/f3b77.png)
![[I^-]](/tpl/images/0549/9170/5a647.png)
![K_{sp}=[Ag^+][I^-]](/tpl/images/0549/9170/9b82b.png)
![[I^-]=\frac{8.3\times 10^{-17}}{2.0\times 10^{-4} M}=4.15\times 10^{-13} M](/tpl/images/0549/9170/c1577.png)
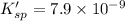
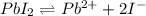
![[Pb^{2+}]=1.5\times 10^{-3} M](/tpl/images/0549/9170/35d28.png)
![K_{sp}'=[Pb^{2+}][I^-]^2](/tpl/images/0549/9170/d5497.png)
![[I^-]^2=\frac{7.9\times 10^{-9}}{1.5\times 10^{-3} M}=4.15\times 10^{-13}](/tpl/images/0549/9170/819e7.png)
![[I^-]=0.0023 M](/tpl/images/0549/9170/2822d.png)


