

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:30
The density of an unknown gas at 98°c and 740 mmhg is 2.50 g/l. what is the molar mass of the gas with work showed?
Answers: 1

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3

Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1
You know the right answer?
Experimental Procedure, Part A.1. Calculate the mass of disodium (molar mass = 372.24 g/mol) requir...
Questions

Mathematics, 11.10.2020 21:01

Mathematics, 11.10.2020 21:01


History, 11.10.2020 21:01

Mathematics, 11.10.2020 21:01


Mathematics, 11.10.2020 21:01

Mathematics, 11.10.2020 21:01


Mathematics, 11.10.2020 21:01

Mathematics, 11.10.2020 21:01


Mathematics, 11.10.2020 21:01


English, 11.10.2020 21:01



Health, 11.10.2020 21:01

Mathematics, 11.10.2020 21:01

Mathematics, 11.10.2020 21:01

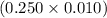 moles = 0.0025 moles
moles = 0.0025 moles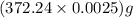 = 0.93 g
= 0.93 g

