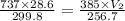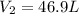A weather balloon is inflated to a volume of 28.6 L at a pressure of 737 mmHg and a temperature of 26.8 ∘C. The balloon rises in the atmosphere to an altitude where the pressure is 385 mmHg and the temperature is -16.3 ∘C. Assuming the balloon can freely expand, calculate the volume of the balloon at this altitude.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1


Chemistry, 23.06.2019 09:30
People who practice which of the following diets may run the risk of not getting enough iron. a. gluten free or vegan diet b. diet for managing diabetes c. vegan diet d. gluten free diet
Answers: 2
You know the right answer?
A weather balloon is inflated to a volume of 28.6 L at a pressure of 737 mmHg and a temperature of 2...
Questions




Mathematics, 04.09.2019 02:30



Mathematics, 04.09.2019 02:30



Spanish, 04.09.2019 02:30



Mathematics, 04.09.2019 02:30



Spanish, 04.09.2019 02:30


Mathematics, 04.09.2019 02:30


Mathematics, 04.09.2019 02:30

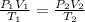
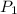 = initial pressure of gas = 737 mm Hg
= initial pressure of gas = 737 mm Hg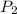 = final pressure of gas = 385 mm Hg
= final pressure of gas = 385 mm Hg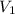 = initial volume of gas = 28.6 L
= initial volume of gas = 28.6 L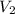 = final volume of gas = ?
= final volume of gas = ?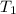 = initial temperature of gas =
= initial temperature of gas = 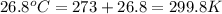
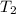 = final temperature of gas =
= final temperature of gas =