Chemistry, 16.03.2020 20:32 hughesbella
Write the cell notation for an electrochemical cell consisting of an anode where Co (s) is oxidized to Co2+ (aq) and a cathode where Fe3+ (aq) is reduced to Fe2+ (aq) at a platinum electrode . Assume all aqueous solutions have a concentration of 1 mol/L and gases have a pressure of 1 bar.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The boiling point of liquids is very high what does it indicate
Answers: 1



Chemistry, 22.06.2019 22:30
Which of the following molecules is polar? c3h7oh c2h5cooh
Answers: 1
You know the right answer?
Write the cell notation for an electrochemical cell consisting of an anode where Co (s) is oxidized...
Questions



Biology, 01.03.2021 23:50



Mathematics, 01.03.2021 23:50

Mathematics, 01.03.2021 23:50


Mathematics, 01.03.2021 23:50


Advanced Placement (AP), 01.03.2021 23:50

Advanced Placement (AP), 01.03.2021 23:50


Chemistry, 01.03.2021 23:50

Mathematics, 01.03.2021 23:50


Chemistry, 01.03.2021 23:50

Mathematics, 01.03.2021 23:50


Mathematics, 01.03.2021 23:50

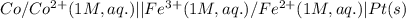
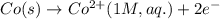
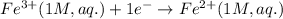 ( × 2)
( × 2)