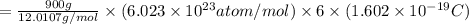How much positive charge is in 0.9 kg of carbon? The atomic weight (12.0107 g) of carbon contains Avogadro’s number of atoms, with each atom having 6 protons and 6 electrons. The elemental charge is 1.602 × 10−19 C and Avogadro’s number is 6.023 × 1023 . Answer in units of C.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:00
What is the cellular process that releases the energy stored in food molecules
Answers: 3

Chemistry, 22.06.2019 01:30
Idon't really understand this can you me and show your work.☺☺[ chemistry b] subject [ electron transfer in lonic bonds]grade( 12)
Answers: 1

Chemistry, 23.06.2019 01:00
What type of chemical bond is formed between two atoms of bromine 1. metallic 2. hydrogen 3. ionic 4. covalent
Answers: 1

Chemistry, 23.06.2019 03:00
Describe the properties of sodium, chlorine, and sodium chloride
Answers: 1
You know the right answer?
How much positive charge is in 0.9 kg of carbon? The atomic weight (12.0107 g) of carbon contains Av...
Questions

Computers and Technology, 06.02.2020 01:48







Social Studies, 06.02.2020 01:49


Mathematics, 06.02.2020 01:49







English, 06.02.2020 01:49


Mathematics, 06.02.2020 01:49

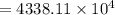 C
C