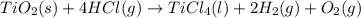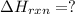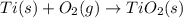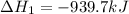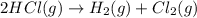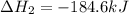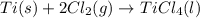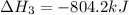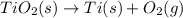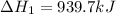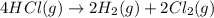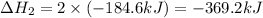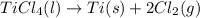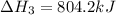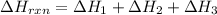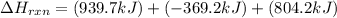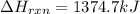Chemistry, 16.03.2020 18:50 kyleighott
Determine the heat of reaction for the process TiO2(s) + 4HCl(g) TiCl4(l) + 2H2(g) + O2(g) using the information given below: Ti(s) + O2(g) TiO2(s) H° = −939.7 kJ 2HCl(g) H2(g) + Cl2(g) H° = −184.6 kJ Ti(s) + 2Cl2(g) TiCl4(l) H° = −804.2 kJ

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:30
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1

Chemistry, 22.06.2019 23:00
What is the most common reason for matter changing its state?
Answers: 1

Chemistry, 23.06.2019 03:30
The semi-conductors on the periodic table are classified as
Answers: 1
You know the right answer?
Determine the heat of reaction for the process TiO2(s) + 4HCl(g) TiCl4(l) + 2H2(g) + O2(g) using the...
Questions



English, 30.01.2020 03:56

Advanced Placement (AP), 30.01.2020 03:56

History, 30.01.2020 03:56

Mathematics, 30.01.2020 03:56






Mathematics, 30.01.2020 03:56





Mathematics, 30.01.2020 03:56

Biology, 30.01.2020 03:56

Mathematics, 30.01.2020 03:56

English, 30.01.2020 03:56