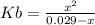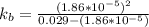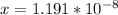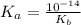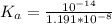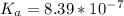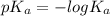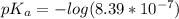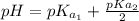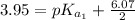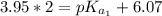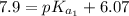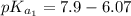Chemistry, 16.03.2020 18:55 winterblanco
The titration of 0.02500 L of a diprotic acid solution with 0.1000 M NaOH requires 34.72 mL of titrant to reach the second equivalence point. The pH is 3.95 at the first equivalence point and 9.27 at the second equivalence point. If the add solution contained 0.2015 g of the acid, what is the molar mass, pK_a1, and pK_a2 of the acid?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:20
Temperature is _related to the average kinetic energy of a gas. inversely directly not disproportionally
Answers: 1

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3


Chemistry, 22.06.2019 23:00
In the reaction h2co3 (aq) + 3nh3 (aq) = 2 nh4+ (aq) + co3 2-, how many electrons are transferred?
Answers: 3
You know the right answer?
The titration of 0.02500 L of a diprotic acid solution with 0.1000 M NaOH requires 34.72 mL of titra...
Questions

Mathematics, 04.12.2020 23:30

English, 04.12.2020 23:30



Mathematics, 04.12.2020 23:30


Physics, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30

Geography, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30




English, 04.12.2020 23:30

Chemistry, 04.12.2020 23:30

Computers and Technology, 04.12.2020 23:30

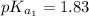
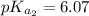
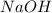 and
and 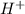 are equal at the equivalence point since they are both taking part in the diprotic acid.
are equal at the equivalence point since they are both taking part in the diprotic acid.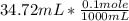
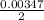 (due to the fact that half of the concentration of NaOH is needed to give the same amount of
(due to the fact that half of the concentration of NaOH is needed to give the same amount of 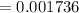
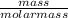
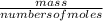
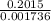
![-log[H^+]](/tpl/images/0548/8178/cbdd4.png)
![[H^+]= 10^{-pH}](/tpl/images/0548/8178/adb7b.png)
![[H^+] = 10^{-9.27](/tpl/images/0548/8178/4fff4.png)
![[H+] = 5.37 * 10^{-10](/tpl/images/0548/8178/27898.png)
![[OH^-]](/tpl/images/0548/8178/b2910.png) can be calculated as follows:
can be calculated as follows:![\frac{10^{-14}}{[H^+]} = \frac {10^{-14}}{5.37* 10^{-10}}](/tpl/images/0548/8178/0ac52.png)
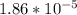
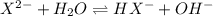
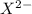 =
= 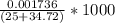
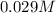

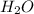
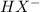
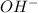
![Kb = \frac{[HX^-][OH^-]}{[X^{2-}]}](/tpl/images/0548/8178/b9431.png)