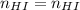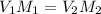Chemistry, 16.03.2020 18:09 TerronRice
You wish to make a 0.299 M hydroiodic acid solution from a stock solution of 6.00 M hydroiodic acid. How much concentrated acid must you add to obtain a total volume of 50.0 mL of the dilute solution?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1

Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

Chemistry, 22.06.2019 15:30
Light waves can move through , but they travel fastest when they move through a(n) .
Answers: 1
You know the right answer?
You wish to make a 0.299 M hydroiodic acid solution from a stock solution of 6.00 M hydroiodic acid....
Questions

Mathematics, 03.12.2021 21:40



Biology, 03.12.2021 21:40

Mathematics, 03.12.2021 21:40



Medicine, 03.12.2021 21:40












History, 03.12.2021 21:50