
Chemistry, 16.03.2020 17:50 RicoCheT89
Aluminum sulfate, known as cake alum, has a wide range of uses, from dyeing leather and cloth to purifying sewage. In aqueous solution, it reacts with base to form a white precipitate. (a) Write balanced total and net ionic equations for its reaction with aqueous NaOH. (b) What mass of precipitate forms when 185.5 mL of 0.533 M NaOH is added to 627 mL of a solution that contains 15.8 g of aluminum sulfate per liter?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Free answer. the treaty of versailles ended world war i, but some of the terms of the treaty contributed to the beginning of world war ii. which was one of the terms of the treaty? the answer would be "germany was forces to pay reparations to the allied countries.". i hope this .
Answers: 1


Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1
You know the right answer?
Aluminum sulfate, known as cake alum, has a wide range of uses, from dyeing leather and cloth to pur...
Questions





History, 25.09.2020 03:01



Mathematics, 25.09.2020 03:01



Mathematics, 25.09.2020 03:01

English, 25.09.2020 03:01

Mathematics, 25.09.2020 03:01




Mathematics, 25.09.2020 03:01

English, 25.09.2020 03:01


English, 25.09.2020 03:01

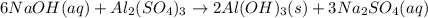
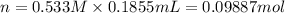
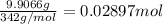
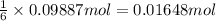
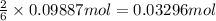 of aluminum hydroxide
of aluminum hydroxide

