
Balance the following chemical equation by the half-reaction method in acidic conditions. Show your work. P4 + NO3–→ H2PO4– + NO(A) What is the oxidized element? What is the reduced element?
(B) Write balanced half reactions for the oxidation reaction:
(C) Write balanced half reactions for the reduction reaction:

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

Chemistry, 22.06.2019 12:30
When a scientific theory has been tested and proved by the scientific community, it becomes a law
Answers: 2

Chemistry, 22.06.2019 17:10
In which block of the periodic table is uranium (u) found? s blockd blockp blockf block
Answers: 1

Chemistry, 22.06.2019 19:30
Helium decays to form lithium. which equation correctly describes this decay?
Answers: 2
You know the right answer?
Balance the following chemical equation by the half-reaction method in acidic conditions. Show your...
Questions

English, 04.04.2020 22:11

Mathematics, 04.04.2020 22:11

Mathematics, 04.04.2020 22:11

Mathematics, 04.04.2020 22:11



Mathematics, 04.04.2020 22:11





Mathematics, 04.04.2020 22:13

History, 04.04.2020 22:14


Mathematics, 04.04.2020 22:14

English, 04.04.2020 22:15




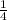 P4 + 4H2O ⇒ H2PO4- + 6H+ + 5e
P4 + 4H2O ⇒ H2PO4- + 6H+ + 5e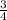 P4 + 5NO3- +2H+ + 2H2O⇒ 5NO+3H3PO4-
P4 + 5NO3- +2H+ + 2H2O⇒ 5NO+3H3PO4-

