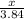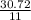Chemistry, 15.03.2020 02:28 poweradampower
The chemical reaction in this example is of environmental interest. Iron pyrite (Fesz) is often an impurity in
coal, and so burning this fuel in a power plant produces sulfur dioxide (SO), a major air pollutant.
Calculate the mass of sulfur dioxide (SO2) produced when 3.84 mol O2 is reacted with Fes, according to the
equation
4 FeSacs + 11 O.
- 2 Fe2O3(-) + S SO2(e)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

Chemistry, 22.06.2019 16:00
Answer asap : ( a. how does mucus prevent the entry of pathogens? b. describe two ways white blood cells protect us from pathogens.
Answers: 1

Chemistry, 23.06.2019 02:00
As light moves from one material into the next, which of the following affects how much the light waves will refract, or bend? angle at which the ray strikes the medium color of the material density of the material temperature of the light wave
Answers: 2
You know the right answer?
The chemical reaction in this example is of environmental interest. Iron pyrite (Fesz) is often an i...
Questions

Mathematics, 17.12.2021 01:00

English, 17.12.2021 01:00



Mathematics, 17.12.2021 01:00

Mathematics, 17.12.2021 01:00

History, 17.12.2021 01:00

Physics, 17.12.2021 01:00

Mathematics, 17.12.2021 01:00

Social Studies, 17.12.2021 01:00

Mathematics, 17.12.2021 01:00

Mathematics, 17.12.2021 01:00

English, 17.12.2021 01:00





English, 17.12.2021 01:00


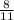 =
=