PLZ HELP, GIVING BRAINLIEST!!
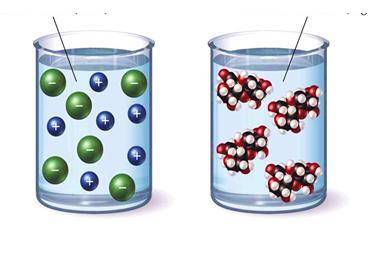
In class, students were given the pictures below and asked to pi...

Chemistry, 14.03.2020 00:29 cxttiemsp021
PLZ HELP, GIVING BRAINLIEST!!
In class, students were given the pictures below and asked to pick a solution that would conduct electricity and to justify their choice.
Based on the model above, which student's argument is correct?
A. Student B claims that the left beaker contains a covalent compound because the solute breaks apart into charged particles.
B. Student D claims to identify the solute as either ionic or covalent more information is needed than what is provided in the model.
C. Student C claims that the right beaker contains an ionic compound because the solute stays together when dissolved.
D. Student A claims that the left beaker contains an ionic compound because the solute breaks apart into charged particles.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Iknow the answer to 13 is b and 14 is d. i just need to know why the correct answers are correct
Answers: 1

Chemistry, 22.06.2019 17:40
Areaction in which products can react to re-form reactants is
Answers: 1

Chemistry, 22.06.2019 20:30
Select all the correct answers.which compounds have the empirical formula ch20? (multiple answers)a.c2h4o2b.c3h603c.ch2o2d.c5h1005e.c6h1206
Answers: 2

Chemistry, 23.06.2019 02:50
Dumbledore decides to gives a surprise demonstration. he starts with a hydrate of na2co3 which has a mass of 4.31 g before heating. after he heats it he finds the mass of the anhydrous compound is found to be 3.22 g. he asks everyone in class to determine the integer x in the hydrate: na2co3·xh2o; you should do this also. round your answer to the nearest integ
Answers: 2
You know the right answer?
Questions








Biology, 10.11.2020 17:00








Mathematics, 10.11.2020 17:00



Mathematics, 10.11.2020 17:00

Biology, 10.11.2020 17:00



