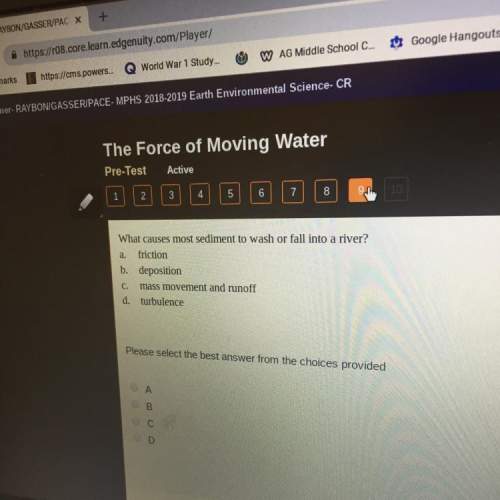
Chemistry, 13.03.2020 19:25 TVASALLO31
The illustration to the left represents a mixture of nitrogen ( blue ) and hydrogen ( light blue ) molecules. If the molecules in the above illustration react to form NH3 according to the equation N2 3 H2 2 NH3 , the limiting reagent is , the number of NH3 molecules formed is , and the number of molecules in excess is..

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:40
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2

Chemistry, 23.06.2019 00:30
•hydration •dissociation •dissolving which one goes to which
Answers: 1

You know the right answer?
The illustration to the left represents a mixture of nitrogen ( blue ) and hydrogen ( light blue ) m...
Questions


Mathematics, 08.01.2021 18:30

History, 08.01.2021 18:30

Mathematics, 08.01.2021 18:30




Business, 08.01.2021 18:30








Mathematics, 08.01.2021 18:30


Mathematics, 08.01.2021 18:30

English, 08.01.2021 18:30

Mathematics, 08.01.2021 18:30