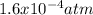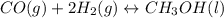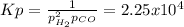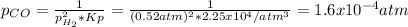Chemistry, 12.03.2020 22:09 demetriascott20
Consider the following reaction, equilibrium concentrations, and equilibrium constant at a particular temperature. Determine the equilibrium pressure of CO. CO(g) + 2 H2(g) CH3OH(l) Kp = 2.25 × 104 P(H2)eq = 0.52 atm

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1

Chemistry, 23.06.2019 01:30
Which statement accurately represents the arrangement of electrons in bohr’s atomic model?
Answers: 2


Chemistry, 23.06.2019 03:30
Name atleast 3 type of energy associated with the microwave
Answers: 1
You know the right answer?
Consider the following reaction, equilibrium concentrations, and equilibrium constant at a particula...
Questions


Engineering, 21.02.2021 19:30


Advanced Placement (AP), 21.02.2021 19:30

Physics, 21.02.2021 19:30


Mathematics, 21.02.2021 19:30


Mathematics, 21.02.2021 19:30



History, 21.02.2021 19:40


Mathematics, 21.02.2021 19:40

Mathematics, 21.02.2021 19:40


History, 21.02.2021 19:40

Mathematics, 21.02.2021 19:40